Project Concordia
A purpose-built agentic AI platform to deliver clinical data harmonization as scalable infrastructure — unlocking the full research potential of patient-donated data.
Patients who participate in clinical trials expect their data to advance future research. But structural interoperability barriers mean much of that data never reaches its full potential.
Project Concordia removes the harmonization bottleneck — delivering an agentic AI platform that transforms months of manual data management into an auditable, reproducible pipeline running in hours. By dramatically reducing cost and complexity, Concordia levels the playing field for under-resourced research teams and ensures shared clinical data achieves the impact patients intended.
AI-Native
Built from the ground up with agentic AI at its core
Multi-Stakeholder
Uniting data contributors, researchers, and platforms
Global Scale
Interoperable infrastructure for clinical research data worldwide
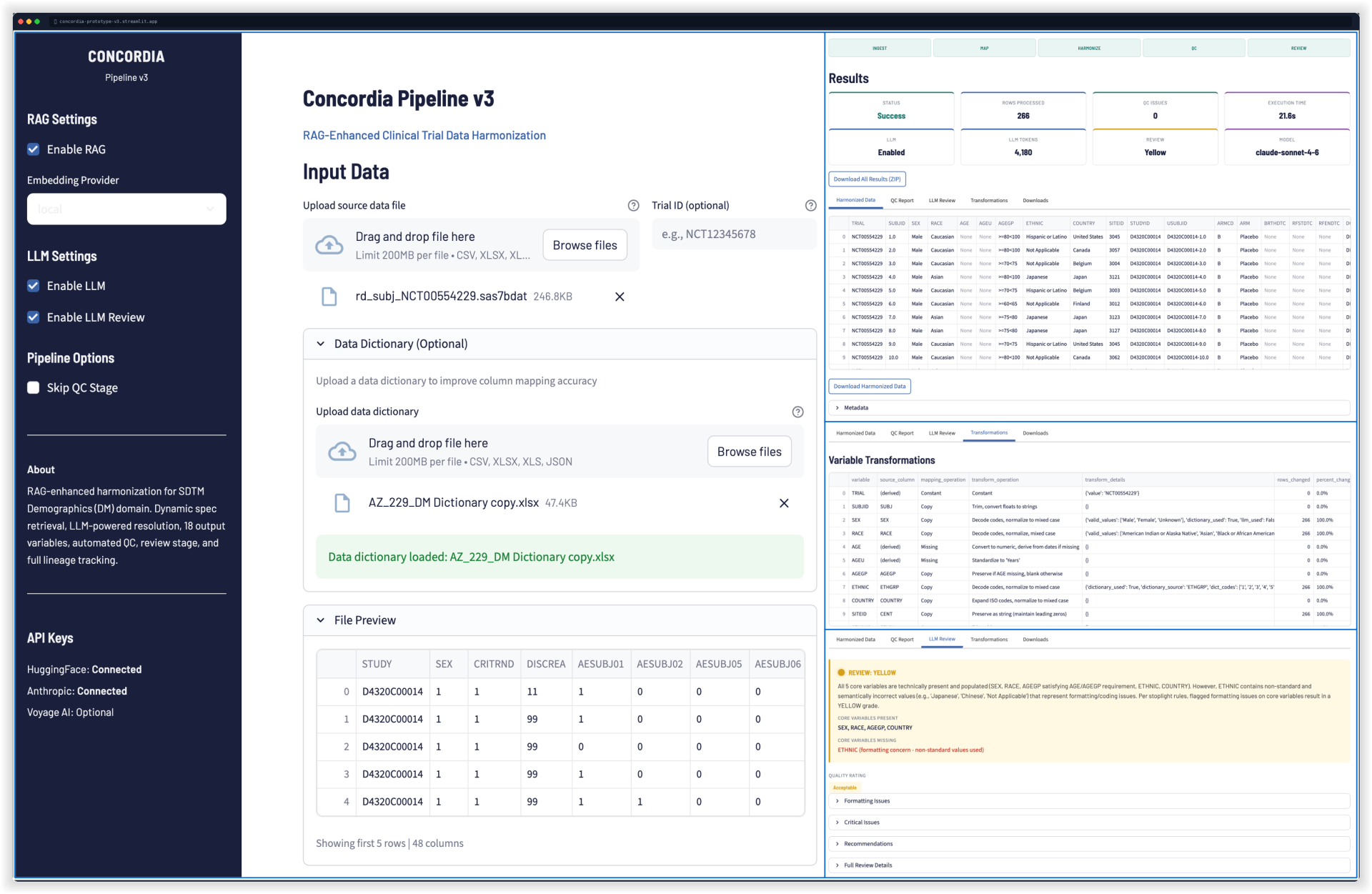
Validated Prototype — Demographics Domain
2026 AI Advisory Communities
CRDSA’s AI-native initiative accelerates AI fluency informed by real-world development, governance, and operational questions.
Our communities are a unique opportunity to accelerate AI fluency informed by real-world development, governance, and operational experiences. Community participation is open to all organizational and individual CRDSA members.
Peer-Driven Community
Bring your expertise and your challenges to a collaborative, peer-driven community
Real-World Insights
Information sharing grounded in real-world use cases and practical experiences
Expert Speakers
Quarterly advisory-specific briefings by community and external AI topic experts
Advisory Symposia
Biannual events sharing the latest AI developments between advisory communities and AI SMEs
Work Sprints & Groups
Initiate work sprints, topic-specific learning communities, and expert working groups
Interested in joining a community?
2022–25 Work Groups
Four focused work groups that established CRDSA’s standards, frameworks, and policy contributions across clinical data sharing.
Innovative Trial Design
Develops new approaches to trial design enhanced by data reuse and drives policy change to facilitate innovative trial execution
Why is this important?
Supplementing RCTs with appropriately sourced Clinical Trial and Real-World Data has the potential to address critical research and trial execution challenges. Effective data reuse has the potential to reduce the trial recruiting burden, improving both the patient experience and sponsor execution efficiency.
What’s been accomplished?
Secondary Use Standards
Establishes common standards and best practices to ensure the integrity, quality, and usability of contributed data
Why is this important?
CRDSA developed these standards in response to common challenges faced by both data contributors and researchers. The standards provide both sponsors and researchers with clear guidelines, ensuring that data is shared appropriately and reused in a way that is fit for purpose.
What’s been accomplished?
- Establishing a Basis for Secondary Use Standards in Clinical Trials (March 2023)
- CRDSA Std 1001: Standard for Sharing Clinical Study Data (September 2024)
- CRDSA Std 2001: Standard for Secondary Analysis of Clinical Study Data (September 2024)
Data Protection
Advances data privacy and governance policies
Why is this important?
The evolving global data protection landscape makes it difficult for many organizations to navigate policy development for internal data reuse and external sharing. This initiative highlights the key issues decision-makers need to consider to develop and implement a data protection policy that balances research access and utility with organization-appropriate risk tolerance.
What’s been accomplished?
- A Review of BioPharma Sponsor Data Sharing Policies and Protection Methodologies (September 2022, updated September 2023)
- Data Protection & Privacy Decision Flow Infographic (February 2024)
Technology & Innovation
Explores new technologies to support novel data governance approaches and advance data sharing principles
Why is this important?
Emerging technologies may enable data sharing initiatives and platforms to develop new governance approaches that advance patient privacy, data utility, and security, facilitating broader access to shared data.
What’s been accomplished?
- Technology Assessment Framework – Excel (November 2022)
- Technology Assessment Framework – R Shiny Application (October 2023)
